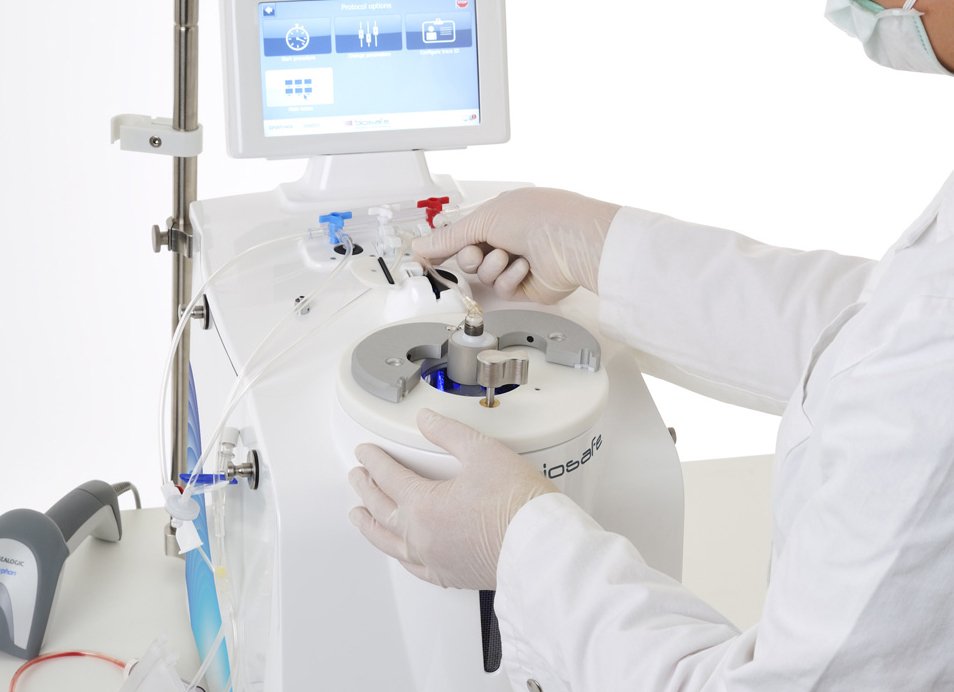
AXP®II Processing
When you store your baby’s stem cells, it is important to understand that Stem Cell processing and isolation is a critical step in cord blood banking. It affects the number of stem cells that can be harvested or recovered from the cord blood. Cell recovery rates are critical because a higher number of stem cells could enhance the success of the transplant or treatment. That is why Cordlife offers FDA-cleared, cGMP and cGTP compliant AXP® II System. This platform is a functionally closed, sterile and automated cord blood processing technology, which uses optical sensor technology to consistently and efficiently harvest the stem cell rich buffy coat and retain over 97% of the Mononuclear cells from Umbilical Cord Blood - All this without using HES, so that you get more stem cells for maximum engraftment and thus better transplant success. The AXP®II consists of the AXP®II device, docking station, processing set, and XpressTRAK® software that assists with cGMP and cGTP compliance.